|
Since its creation, the standards organization has become a recognized creator of good practice worldwide. GAMP also sought to encourage understanding of how computer systems validation should be handled in the pharmaceutical industry. This included improved working relationships with agencies in other countries like the United States Food and Drug Administration ( FDA). GAMP was created in 1991 in the United Kingdom to address the pharmaceutical industry’s desire to create guidelines that would improve understanding of changing expectations of regulatory agencies in Europe. The document provides an outline for the application of risk management principles and tools for computer system validation. One of the most guidance documents is GAMP 5 Guide: Compliant GxP Computerized Systems, which is often simply referred to as GAMP 5. GAMP publications include practice guides on calibration management, compliance, IT infrastructure, data archiving and process validation. Category 4 Configured software including, LIMS, SCADA, DCS, CDS, etc. Category 3 Non configurable software including, commercial off the shelf software (COTS), Laboratory Instruments / Software. This rigorous employment of standard of operating procedures (SOP) helps ensure purity of manufactured pharmaceuticals. Advertisement GAMP Categories Category 1 Infrastructure software including operating systems, Database Managers, etc. Rather than testing quality control batches, GAMP makes quality testing an integral part of each stage of manufacturing, including facilities, equipment, materials acquisition and staff hygiene. 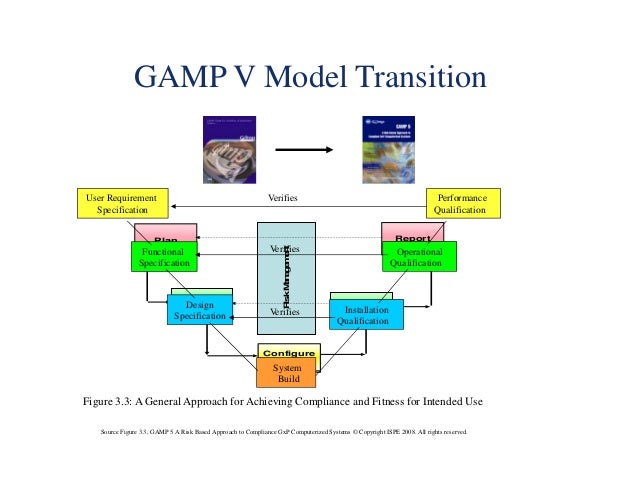
GAMP COP (Community of Practice) is a pharmaceutical professionals’ forum that ensures continued development and adoption of best practices in the field. 
However, when working with commercial off-the-shelf software, (Category. GAMP guidelines are used heavily by the pharmaceutical industry to ensure that drugs are manufactured with the required quality. GAMP is an acronym for Good Automated Manufacturing Practice, (which is hopefully quite familiar to you), for a risk-based approach to computer system validation where a system is evaluated and assigned to a predefined category based on its intended use and complexity. GAMP is also a subcommittee of the International Society for Pharmaceutical Engineering (ISPE). Good automated manufacturing practice (GAMP) is a set of guidelines for manufacturers and other automation users follow to maintain operational efficiency and reliability.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
